2025
Leveraging massive H&E morphological models for phase contrast microscopy images with a generative deep learning approach
Zhi Fei Dong, Roya Navab, Michael Cabanero, Irene Xie, Ming-Sound Tsao, Chris McIntosh, Gregory W. Schwartz# (# corresponding author)
Submitted to Light: Science and Applications; Under review. 2025
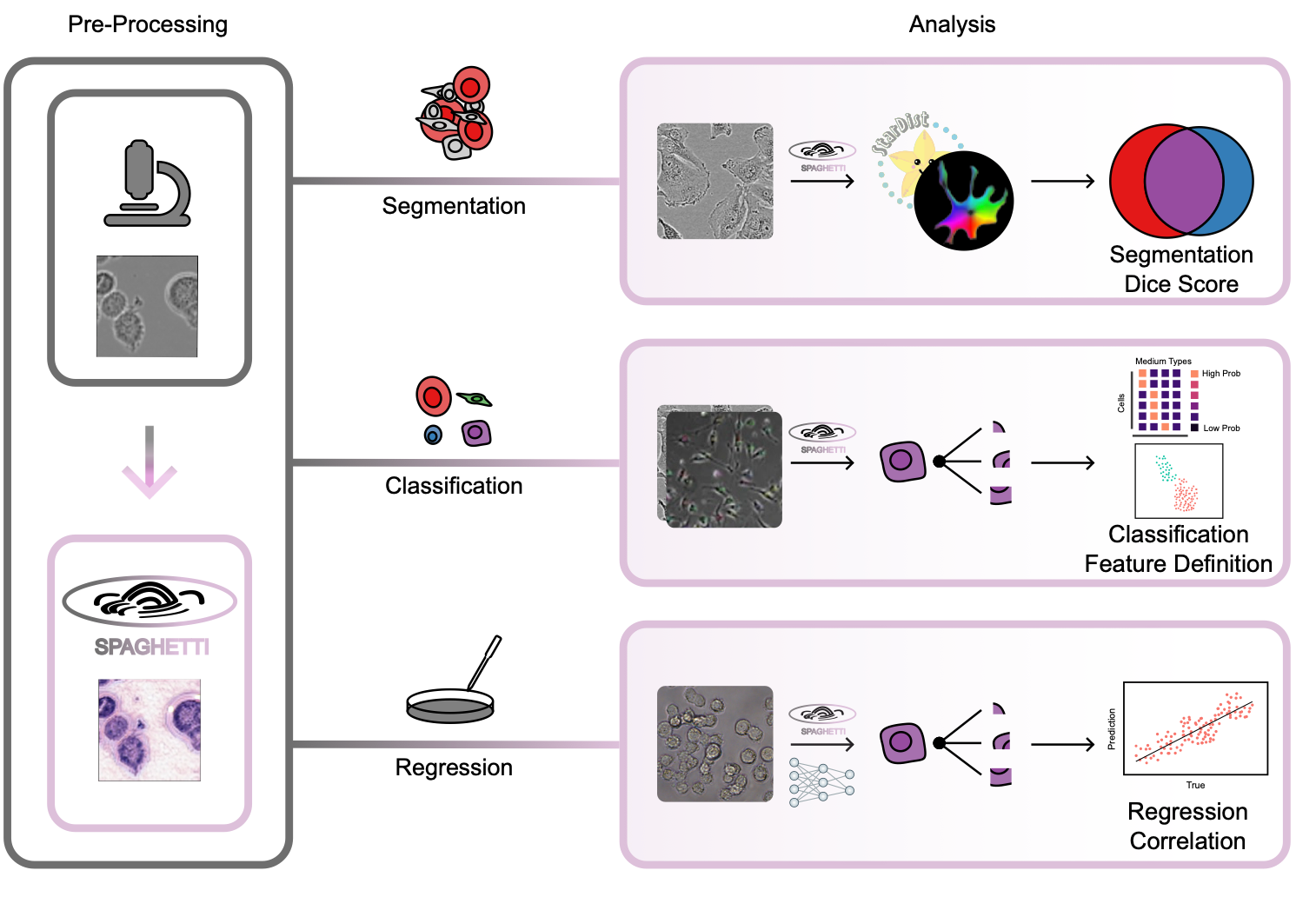
Phase contrast microscopy (PCM) is a powerful cell imaging method, and one of the few technologies to delineate and track cell structure in live cells without staining. Despite PCM’s great potential and popularity in monitoring live-cell populations, there are few algorithms to extract morphological information from these images due to the lack of large training datasets. To overcome this challenge and enable advanced, high- throughput, quantitative analysis of PCM images, we introduce SPAGHETTI (Structural Phase Adaptation via Generative Histological Enhancement and Texture-preserving Translation Integration): a lightweight image preprocessor built on a modified cycle-consistent generative adversarial network. SPAGHETTI preprocessing enables the use of large-scale deep learning models, originally trained on hematoxylin and eosin (H&E) pathological images that are pervasive and widespread in clinical settings, on PCM images through style translation. We demonstrate that using SPAGHETTI pre- processing achieves significantly improves performance on cell segmentation through the use of tissue and H&E-specific cell segmentation models. We also show that by passing translated PCM images across several independent datasets into H&E feature extractor models, we improve the performance of cell-type annotation, experimental media classification, and cell viability prediction. Overall, SPAGHETTI enables many general quantitative analyses of PCM that were previously difficult to perform due to the dearth of PCM-specific feature extractors, and acts as a valuable preprocessing step to gather new cell-state information through downstream analysis of morphological features.
Leveraging massive H&E morphological models for phase contrast microscopy images with a generative deep learning approach
Zhi Fei Dong, Roya Navab, Michael Cabanero, Irene Xie, Ming-Sound Tsao, Chris McIntosh, Gregory W. Schwartz# (# corresponding author)
Submitted to Light: Science and Applications; Under review. 2025
Phase contrast microscopy (PCM) is a powerful cell imaging method, and one of the few technologies to delineate and track cell structure in live cells without staining. Despite PCM’s great potential and popularity in monitoring live-cell populations, there are few algorithms to extract morphological information from these images due to the lack of large training datasets. To overcome this challenge and enable advanced, high- throughput, quantitative analysis of PCM images, we introduce SPAGHETTI (Structural Phase Adaptation via Generative Histological Enhancement and Texture-preserving Translation Integration): a lightweight image preprocessor built on a modified cycle-consistent generative adversarial network. SPAGHETTI preprocessing enables the use of large-scale deep learning models, originally trained on hematoxylin and eosin (H&E) pathological images that are pervasive and widespread in clinical settings, on PCM images through style translation. We demonstrate that using SPAGHETTI pre- processing achieves significantly improves performance on cell segmentation through the use of tissue and H&E-specific cell segmentation models. We also show that by passing translated PCM images across several independent datasets into H&E feature extractor models, we improve the performance of cell-type annotation, experimental media classification, and cell viability prediction. Overall, SPAGHETTI enables many general quantitative analyses of PCM that were previously difficult to perform due to the dearth of PCM-specific feature extractors, and acts as a valuable preprocessing step to gather new cell-state information through downstream analysis of morphological features.
2023
Going below and beyond the surface: Microneedle structure, materials, drugs, fabrication, and applications for wound healing and tissue regeneration
Shang Lyu, Zhifei Dong, Xiaoxiao Xu, Ho-Pan Bei, Ho-Yin Yuen, Chung-Wai James Cheung, Man-Sang Wong, Yong He, Xin Zhao
Bioactive Materials 2023
Microneedle, as a novel drug delivery system, has attracted widespread attention due to its non-invasiveness, painless and simple administration, controllable drug delivery, and diverse cargo loading capacity. Although microneedles are initially designed to penetrate stratum corneum of skin for transdermal drug delivery, they, recently, have been used to promote wound healing and regeneration of diverse tissues and organs and the results are promising. Despite there are reviews about microneedles, few of them focus on wound healing and tissue regeneration. Here, we review the recent advances of microneedles in this field. We first give an overview of microneedle system in terms of its potential cargos (e.g., small molecules, macromolecules, nucleic acids, nanoparticles, extracellular vesicle, cells), structural designs (e.g., multidrug structures, adhesive structures), material selection, and drug release mechanisms. Then we briefly summarize different microneedle fabrication methods, including their advantages and limitations. We finally summarize the recent progress of microneedle-assisted wound healing and tissue regeneration (e.g., skin, cardiac, bone, tendon, ocular, vascular, oral, hair, spinal cord, and uterine tissues). We expect that our article would serve as a guideline for readers to design their microneedle systems according to different applications, including material selection, drug selection, and structure design, for achieving better healing and regeneration efficacy.
Going below and beyond the surface: Microneedle structure, materials, drugs, fabrication, and applications for wound healing and tissue regeneration
Shang Lyu, Zhifei Dong, Xiaoxiao Xu, Ho-Pan Bei, Ho-Yin Yuen, Chung-Wai James Cheung, Man-Sang Wong, Yong He, Xin Zhao
Bioactive Materials 2023
Microneedle, as a novel drug delivery system, has attracted widespread attention due to its non-invasiveness, painless and simple administration, controllable drug delivery, and diverse cargo loading capacity. Although microneedles are initially designed to penetrate stratum corneum of skin for transdermal drug delivery, they, recently, have been used to promote wound healing and regeneration of diverse tissues and organs and the results are promising. Despite there are reviews about microneedles, few of them focus on wound healing and tissue regeneration. Here, we review the recent advances of microneedles in this field. We first give an overview of microneedle system in terms of its potential cargos (e.g., small molecules, macromolecules, nucleic acids, nanoparticles, extracellular vesicle, cells), structural designs (e.g., multidrug structures, adhesive structures), material selection, and drug release mechanisms. Then we briefly summarize different microneedle fabrication methods, including their advantages and limitations. We finally summarize the recent progress of microneedle-assisted wound healing and tissue regeneration (e.g., skin, cardiac, bone, tendon, ocular, vascular, oral, hair, spinal cord, and uterine tissues). We expect that our article would serve as a guideline for readers to design their microneedle systems according to different applications, including material selection, drug selection, and structure design, for achieving better healing and regeneration efficacy.
2022
Evaporation-based, co-axial lock-and-key fibrous reservoir for long-term prevention of hypertrophic scars
Ho-Pan Bei, Tianpeng Xu, Jing Zhou, Zhifei Dong, Yufeng Wang, Kak-Yuen Wong, Huaiyu Wang, Xin Zhao
Applied Materials Today 2022
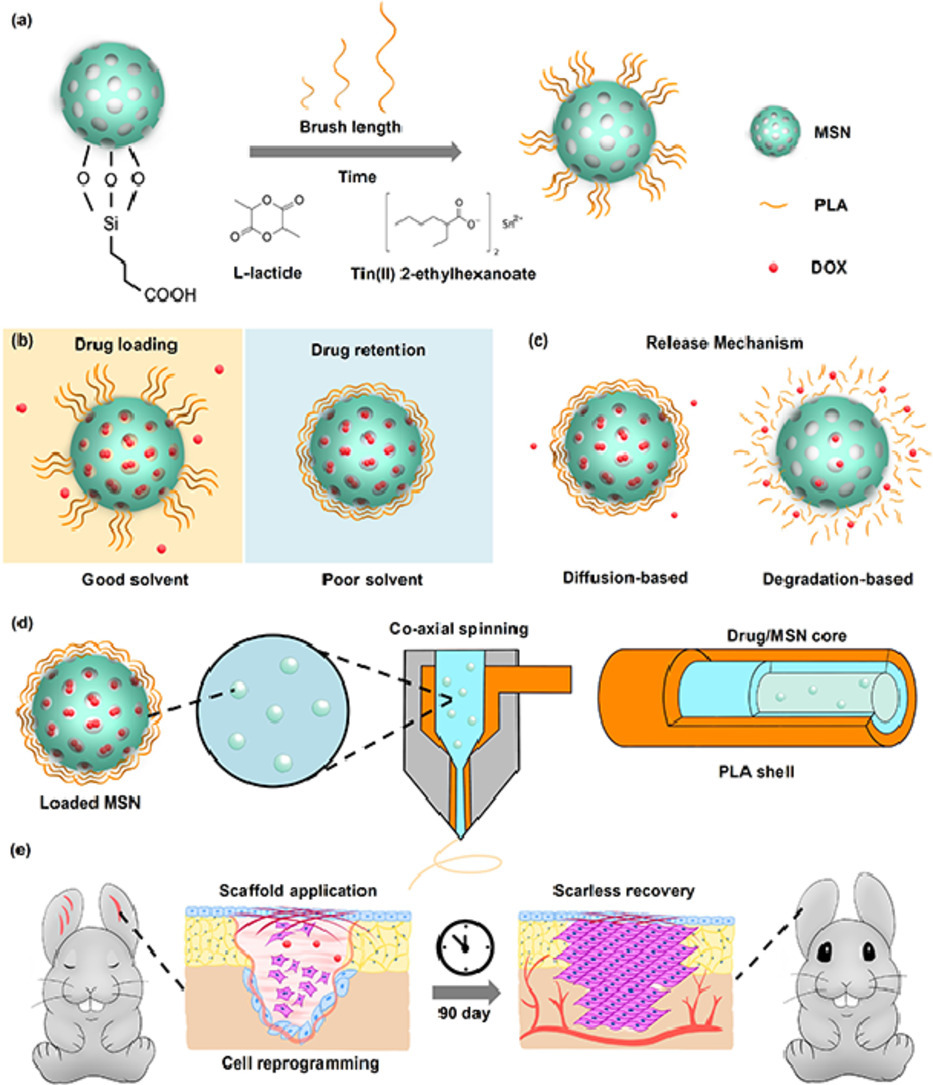
Many diseases and conditions such as hypertrophic scarring require long-term maintenance over the healing cycle to achieve full recovery. However, there is a lack of wound dressings that can sustain over 90 days of therapeutic release. Inspired by the enhancement of wound healing by the nanofibrous morphology and diverse structures of electrospinning, we report an evaporation-based co-axial electrospun fibrous scaffold incorporating polymer brush gatekept nanocarriers for sustained delivery of therapeutics. The release rates of the system were demonstrated to be tunable through polymer graft length, while the system experienced minimal burst release when submerged under aqueous conditions. As a proof-of-concept, we target hypertrophic scarring by loading the system with doxorubicin, which led to inhibition of fibroblast activity without interfering with cell adhesion. Application of our scaffolds on rabbit ear hypertrophic scar models displayed that our scaffolds effectively reduced collagen density and scar-related gene expression in healing tissues, with improved tissue elevation outcomes. We envision that our long-term release scaffolds will be useful in combating long unresolved clinical dilemma such as tendon adhesion and tumor regression.
Evaporation-based, co-axial lock-and-key fibrous reservoir for long-term prevention of hypertrophic scars
Ho-Pan Bei, Tianpeng Xu, Jing Zhou, Zhifei Dong, Yufeng Wang, Kak-Yuen Wong, Huaiyu Wang, Xin Zhao
Applied Materials Today 2022
Many diseases and conditions such as hypertrophic scarring require long-term maintenance over the healing cycle to achieve full recovery. However, there is a lack of wound dressings that can sustain over 90 days of therapeutic release. Inspired by the enhancement of wound healing by the nanofibrous morphology and diverse structures of electrospinning, we report an evaporation-based co-axial electrospun fibrous scaffold incorporating polymer brush gatekept nanocarriers for sustained delivery of therapeutics. The release rates of the system were demonstrated to be tunable through polymer graft length, while the system experienced minimal burst release when submerged under aqueous conditions. As a proof-of-concept, we target hypertrophic scarring by loading the system with doxorubicin, which led to inhibition of fibroblast activity without interfering with cell adhesion. Application of our scaffolds on rabbit ear hypertrophic scar models displayed that our scaffolds effectively reduced collagen density and scar-related gene expression in healing tissues, with improved tissue elevation outcomes. We envision that our long-term release scaffolds will be useful in combating long unresolved clinical dilemma such as tendon adhesion and tumor regression.
Biomimicking design of artificial periosteum for promoting bone healing
Yuhe Yang, Jingdong Rao, Huaqian Liu, Zhifei Dong, Zhen Zhang, Ho-Pan Bei, Chunyi Wen, Xin Zhao
Journal of Orthopaedic Translation 2022
Background Periosteum is a vascularized tissue membrane covering the bone surface and plays a decisive role in bone reconstruction process after fracture. Various artificial periosteum has been developed to assist the allografts or bionic bone scaffolds in accelerating bone healing. Recently, the biomimicking design of artificial periosteum has attracted increasing attention due to the recapitulation of the natural extracellular microenvironment of the periosteum and has presented unique capacity to modulate the cell fates and ultimately enhance the bone formation and improve neovascularization. Methods A systematic literature search is performed and relevant findings in biomimicking design of artificial periosteum have been reviewed and cited. Results We give a systematical overview of current development of biomimicking design of artificial periosteum. We first summarize the universal strategies for designing biomimicking artificial periosteum including biochemical biomimicry and biophysical biomimicry aspects. We then discuss three types of novel versatile biomimicking artificial periosteum including physical-chemical combined artificial periosteum, heterogeneous structured biomimicking periosteum, and healing phase-targeting biomimicking periosteum. Finally, we comment on the potential implications and prospects in the future design of biomimicking artificial periosteum. Conclusion This review summarizes the preparation strategies of biomimicking artificial periosteum in recent years with a discussion of material selection, animal model adoption, biophysical and biochemical cues to regulate the cell fates as well as three types of latest developed versatile biomimicking artificial periosteum. In future, integration of innervation, osteochondral regeneration, and osteoimmunomodulation, should be taken into consideration when fabricating multifunctional artificial periosteum. The Translational Potential of this Article: This study provides a holistic view on the design strategy and the therapeutic potential of biomimicking artificial periosteum to promote bone healing. It is hoped to open a new avenue of artificial periosteum design with biomimicking considerations and reposition of the current strategy for accelerated bone healing.
Biomimicking design of artificial periosteum for promoting bone healing
Yuhe Yang, Jingdong Rao, Huaqian Liu, Zhifei Dong, Zhen Zhang, Ho-Pan Bei, Chunyi Wen, Xin Zhao
Journal of Orthopaedic Translation 2022
Background Periosteum is a vascularized tissue membrane covering the bone surface and plays a decisive role in bone reconstruction process after fracture. Various artificial periosteum has been developed to assist the allografts or bionic bone scaffolds in accelerating bone healing. Recently, the biomimicking design of artificial periosteum has attracted increasing attention due to the recapitulation of the natural extracellular microenvironment of the periosteum and has presented unique capacity to modulate the cell fates and ultimately enhance the bone formation and improve neovascularization. Methods A systematic literature search is performed and relevant findings in biomimicking design of artificial periosteum have been reviewed and cited. Results We give a systematical overview of current development of biomimicking design of artificial periosteum. We first summarize the universal strategies for designing biomimicking artificial periosteum including biochemical biomimicry and biophysical biomimicry aspects. We then discuss three types of novel versatile biomimicking artificial periosteum including physical-chemical combined artificial periosteum, heterogeneous structured biomimicking periosteum, and healing phase-targeting biomimicking periosteum. Finally, we comment on the potential implications and prospects in the future design of biomimicking artificial periosteum. Conclusion This review summarizes the preparation strategies of biomimicking artificial periosteum in recent years with a discussion of material selection, animal model adoption, biophysical and biochemical cues to regulate the cell fates as well as three types of latest developed versatile biomimicking artificial periosteum. In future, integration of innervation, osteochondral regeneration, and osteoimmunomodulation, should be taken into consideration when fabricating multifunctional artificial periosteum. The Translational Potential of this Article: This study provides a holistic view on the design strategy and the therapeutic potential of biomimicking artificial periosteum to promote bone healing. It is hoped to open a new avenue of artificial periosteum design with biomimicking considerations and reposition of the current strategy for accelerated bone healing.
Shedding light on 3D printing: Printing photo-crosslinkable constructs for tissue engineering
Qiang Zhang, Ho-Pan Bei, Mengna Zhao, Zhifei Dong, Xin Zhao
Journal of Orthopaedic Translation 2022
3D printing has emerged as a pivotal fabrication technique for preparing scaffolds for engineering tissues and tissue models. Among different 3D printing platforms, photo-crosslinking-based 3D printing techniques like digital light processing and stereolithography have become most popular as they enable the construction of complex architecture with improved spatial resolution, reliable pattern fidelity, and high printing speed. In addition, by selecting appropriate ink combinations or modulating the photo-crosslinking printing parameters (e.g., the types or concentrations of photoinitiators and crosslinkers, light exposure time or intensity, as well as the 3D printing techniques used), the structures and properties (e.g., swelling and mechanical properties) of the resultant printed scaffolds can be finely tailored to meet the practical application requirements. Here, recent advances on the promising development of photo-crosslinkable materials for 3D printing with a focus on their biomedical applications for repairing damaged organs and developing in vitro tissue models are reviewed. Firstly, an overview of commonly used photo-crosslinkable materials, as well as insights on how the printing outcomes of these materials can be improved are provided. Then, the diverse regulation strategies of the photo-polymerization process and the 3D printing parameters to improve the performances of the printed structures are summarized. The existing challenges and future directions are finally discussed from the technical and application perspectives of photo-crosslinking-based 3D printing.
Shedding light on 3D printing: Printing photo-crosslinkable constructs for tissue engineering
Qiang Zhang, Ho-Pan Bei, Mengna Zhao, Zhifei Dong, Xin Zhao
Journal of Orthopaedic Translation 2022
3D printing has emerged as a pivotal fabrication technique for preparing scaffolds for engineering tissues and tissue models. Among different 3D printing platforms, photo-crosslinking-based 3D printing techniques like digital light processing and stereolithography have become most popular as they enable the construction of complex architecture with improved spatial resolution, reliable pattern fidelity, and high printing speed. In addition, by selecting appropriate ink combinations or modulating the photo-crosslinking printing parameters (e.g., the types or concentrations of photoinitiators and crosslinkers, light exposure time or intensity, as well as the 3D printing techniques used), the structures and properties (e.g., swelling and mechanical properties) of the resultant printed scaffolds can be finely tailored to meet the practical application requirements. Here, recent advances on the promising development of photo-crosslinkable materials for 3D printing with a focus on their biomedical applications for repairing damaged organs and developing in vitro tissue models are reviewed. Firstly, an overview of commonly used photo-crosslinkable materials, as well as insights on how the printing outcomes of these materials can be improved are provided. Then, the diverse regulation strategies of the photo-polymerization process and the 3D printing parameters to improve the performances of the printed structures are summarized. The existing challenges and future directions are finally discussed from the technical and application perspectives of photo-crosslinking-based 3D printing.
2021
Application of TPMS structure in bone regeneration
Zhifei Dong, Xin Zhao
Engineered Regeneration 2021
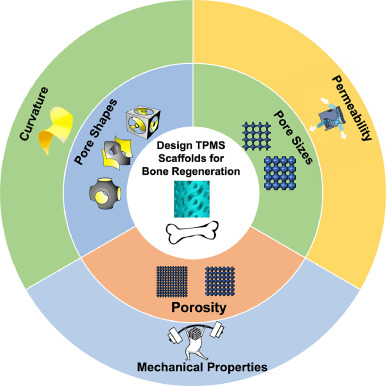
Bone defect repair, due to its complex process in nature, has become a costly issue in modern day medicine. This causes a growing demand for a bone substitute that is effective and easy to construct. Recently, triply periodic minimal surface (TPMS) scaffolds, which embody trabecular bone-mimicking hyperboloidal topography, have become a promising candidate for this exact role due to their unique structure to promote many cellular processes. In response to the growing popularity of TPMS scaffolds amongst researchers, this review discusses the effect of different parameters (including pore size, porosity, and pore shape, as well as their influences on mechanical property, permeability, and curvature), along with the controlling and designing of such parameters, on bone regeneration to serve as a guide for future researchers in designing and utilizing TPMS scaffolds for bone regeneration purposes.
Application of TPMS structure in bone regeneration
Zhifei Dong, Xin Zhao
Engineered Regeneration 2021
Bone defect repair, due to its complex process in nature, has become a costly issue in modern day medicine. This causes a growing demand for a bone substitute that is effective and easy to construct. Recently, triply periodic minimal surface (TPMS) scaffolds, which embody trabecular bone-mimicking hyperboloidal topography, have become a promising candidate for this exact role due to their unique structure to promote many cellular processes. In response to the growing popularity of TPMS scaffolds amongst researchers, this review discusses the effect of different parameters (including pore size, porosity, and pore shape, as well as their influences on mechanical property, permeability, and curvature), along with the controlling and designing of such parameters, on bone regeneration to serve as a guide for future researchers in designing and utilizing TPMS scaffolds for bone regeneration purposes.