

My name is Richard Dong, a Ph.D. student supervised by Dr. Gregory Schwartz in the Department of Medical Biophysics at the University of Toronto, Toronto, Ontario, Canada. I am working on developing machine learning algorithms for live cell imaging analysis. I majored in Medical Physics and minored in Biology and Bioinformatics during my undergraduate study at the University of Waterloo, and am excited about how to utilize biological data to answer big questions in biology.
I used to live in Shanghai, China, and moved to Toronto, Canada when I was 14 years old.
Warning
Problem: The current name of your GitHub Pages repository ("Solution: Please consider renaming the repository to "
http://".
However, if the current repository name is intended, you can ignore this message by removing "{% include widgets/debug_repo_name.html %}" in index.html.
Action required
Problem: The current root path of this site is "baseurl ("_config.yml.
Solution: Please set the
baseurl in _config.yml to "Education
-
 University of TorontoDepartment of Medical Biophysics, Temerty Faculty of Medicine
University of TorontoDepartment of Medical Biophysics, Temerty Faculty of Medicine
Ph.D. CandidateSep. 2023 - present -
 University of WaterlooB.Sc. (Honours) in Medical Physics, Minor in Biology and BioinformaticsSep. 2018 - Jun. 2023
University of WaterlooB.Sc. (Honours) in Medical Physics, Minor in Biology and BioinformaticsSep. 2018 - Jun. 2023
Affiliations
-
Ph.D. Candidate, University of TorontoSep. 2025
-
Graduate Researcher, Princess Margaret Cancer Centre, University Health NetworkJan. 2024
-
Faculty Affiliated Researcher, Vector Institute of Artificial IntelligenceSep. 2024
Selected Honours & Awards
-
STARS21 Scholarship2025
-
Ontario Graduate Scholarship (OGS)2024
-
Temerty Faculty of Medicine Entrance Scholarship (Top 10 applicants)2023
News
Selected Publications (view all )
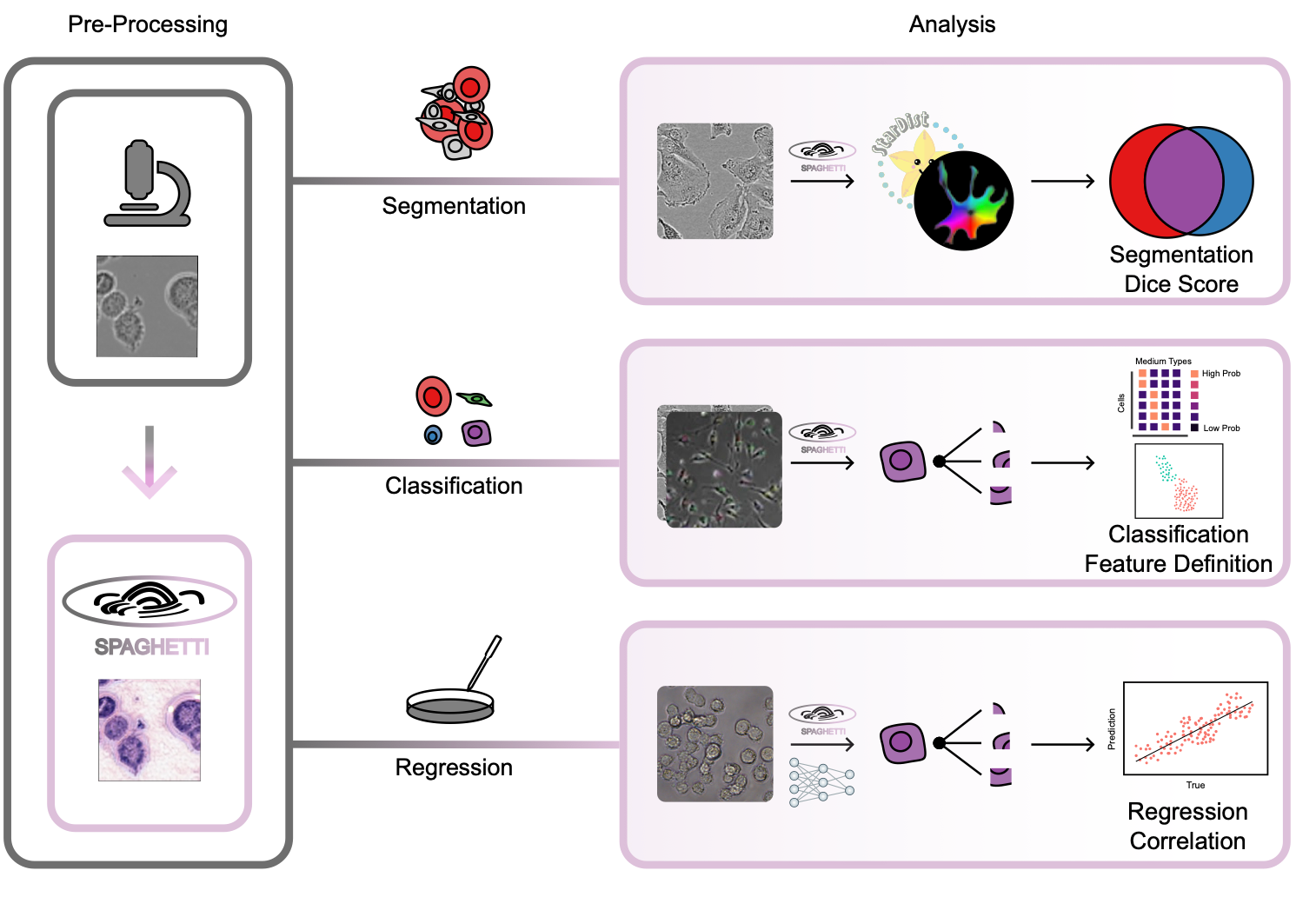
Leveraging massive H&E morphological models for phase contrast microscopy images with a generative deep learning approach
Zhi Fei Dong, Roya Navab, Michael Cabanero, Irene Xie, Ming-Sound Tsao, Chris McIntosh, Gregory W. Schwartz# (# corresponding author)
Submitted to Light: Science and Applications; Under review. 2025
Phase contrast microscopy (PCM) is a powerful cell imaging method, and one of the few technologies to delineate and track cell structure in live cells without staining. Despite PCM’s great potential and popularity in monitoring live-cell populations, there are few algorithms to extract morphological information from these images due to the lack of large training datasets. To overcome this challenge and enable advanced, high- throughput, quantitative analysis of PCM images, we introduce SPAGHETTI (Structural Phase Adaptation via Generative Histological Enhancement and Texture-preserving Translation Integration): a lightweight image preprocessor built on a modified cycle-consistent generative adversarial network. SPAGHETTI preprocessing enables the use of large-scale deep learning models, originally trained on hematoxylin and eosin (H&E) pathological images that are pervasive and widespread in clinical settings, on PCM images through style translation. We demonstrate that using SPAGHETTI pre- processing achieves significantly improves performance on cell segmentation through the use of tissue and H&E-specific cell segmentation models. We also show that by passing translated PCM images across several independent datasets into H&E feature extractor models, we improve the performance of cell-type annotation, experimental media classification, and cell viability prediction. Overall, SPAGHETTI enables many general quantitative analyses of PCM that were previously difficult to perform due to the dearth of PCM-specific feature extractors, and acts as a valuable preprocessing step to gather new cell-state information through downstream analysis of morphological features.
Leveraging massive H&E morphological models for phase contrast microscopy images with a generative deep learning approach
Zhi Fei Dong, Roya Navab, Michael Cabanero, Irene Xie, Ming-Sound Tsao, Chris McIntosh, Gregory W. Schwartz# (# corresponding author)
Submitted to Light: Science and Applications; Under review. 2025
Phase contrast microscopy (PCM) is a powerful cell imaging method, and one of the few technologies to delineate and track cell structure in live cells without staining. Despite PCM’s great potential and popularity in monitoring live-cell populations, there are few algorithms to extract morphological information from these images due to the lack of large training datasets. To overcome this challenge and enable advanced, high- throughput, quantitative analysis of PCM images, we introduce SPAGHETTI (Structural Phase Adaptation via Generative Histological Enhancement and Texture-preserving Translation Integration): a lightweight image preprocessor built on a modified cycle-consistent generative adversarial network. SPAGHETTI preprocessing enables the use of large-scale deep learning models, originally trained on hematoxylin and eosin (H&E) pathological images that are pervasive and widespread in clinical settings, on PCM images through style translation. We demonstrate that using SPAGHETTI pre- processing achieves significantly improves performance on cell segmentation through the use of tissue and H&E-specific cell segmentation models. We also show that by passing translated PCM images across several independent datasets into H&E feature extractor models, we improve the performance of cell-type annotation, experimental media classification, and cell viability prediction. Overall, SPAGHETTI enables many general quantitative analyses of PCM that were previously difficult to perform due to the dearth of PCM-specific feature extractors, and acts as a valuable preprocessing step to gather new cell-state information through downstream analysis of morphological features.
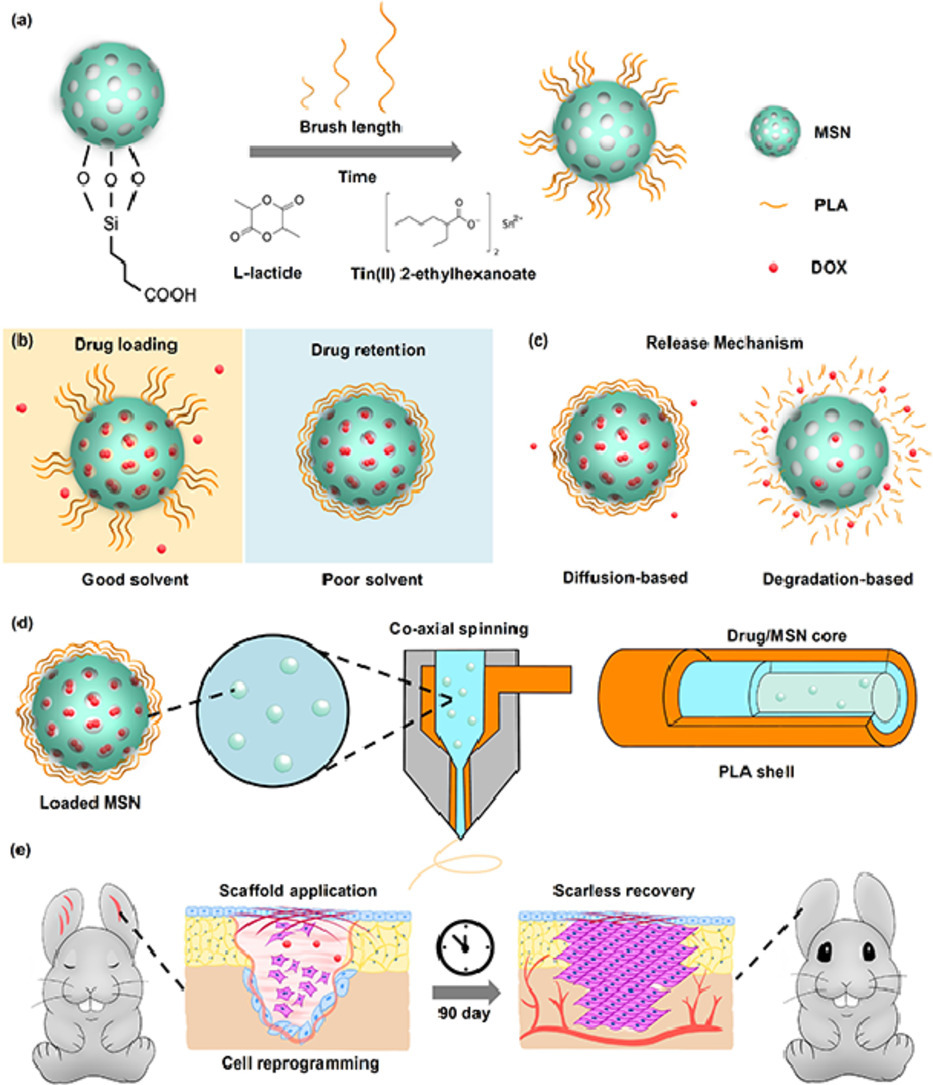
Evaporation-based, co-axial lock-and-key fibrous reservoir for long-term prevention of hypertrophic scars
Ho-Pan Bei, Tianpeng Xu, Jing Zhou, Zhifei Dong, Yufeng Wang, Kak-Yuen Wong, Huaiyu Wang, Xin Zhao
Applied Materials Today 2022
Many diseases and conditions such as hypertrophic scarring require long-term maintenance over the healing cycle to achieve full recovery. However, there is a lack of wound dressings that can sustain over 90 days of therapeutic release. Inspired by the enhancement of wound healing by the nanofibrous morphology and diverse structures of electrospinning, we report an evaporation-based co-axial electrospun fibrous scaffold incorporating polymer brush gatekept nanocarriers for sustained delivery of therapeutics. The release rates of the system were demonstrated to be tunable through polymer graft length, while the system experienced minimal burst release when submerged under aqueous conditions. As a proof-of-concept, we target hypertrophic scarring by loading the system with doxorubicin, which led to inhibition of fibroblast activity without interfering with cell adhesion. Application of our scaffolds on rabbit ear hypertrophic scar models displayed that our scaffolds effectively reduced collagen density and scar-related gene expression in healing tissues, with improved tissue elevation outcomes. We envision that our long-term release scaffolds will be useful in combating long unresolved clinical dilemma such as tendon adhesion and tumor regression.
Evaporation-based, co-axial lock-and-key fibrous reservoir for long-term prevention of hypertrophic scars
Ho-Pan Bei, Tianpeng Xu, Jing Zhou, Zhifei Dong, Yufeng Wang, Kak-Yuen Wong, Huaiyu Wang, Xin Zhao
Applied Materials Today 2022
Many diseases and conditions such as hypertrophic scarring require long-term maintenance over the healing cycle to achieve full recovery. However, there is a lack of wound dressings that can sustain over 90 days of therapeutic release. Inspired by the enhancement of wound healing by the nanofibrous morphology and diverse structures of electrospinning, we report an evaporation-based co-axial electrospun fibrous scaffold incorporating polymer brush gatekept nanocarriers for sustained delivery of therapeutics. The release rates of the system were demonstrated to be tunable through polymer graft length, while the system experienced minimal burst release when submerged under aqueous conditions. As a proof-of-concept, we target hypertrophic scarring by loading the system with doxorubicin, which led to inhibition of fibroblast activity without interfering with cell adhesion. Application of our scaffolds on rabbit ear hypertrophic scar models displayed that our scaffolds effectively reduced collagen density and scar-related gene expression in healing tissues, with improved tissue elevation outcomes. We envision that our long-term release scaffolds will be useful in combating long unresolved clinical dilemma such as tendon adhesion and tumor regression.
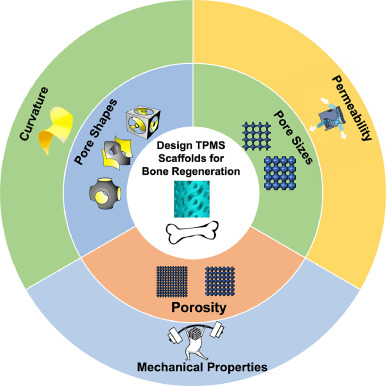
Application of TPMS structure in bone regeneration
Zhifei Dong, Xin Zhao
Engineered Regeneration 2021
Bone defect repair, due to its complex process in nature, has become a costly issue in modern day medicine. This causes a growing demand for a bone substitute that is effective and easy to construct. Recently, triply periodic minimal surface (TPMS) scaffolds, which embody trabecular bone-mimicking hyperboloidal topography, have become a promising candidate for this exact role due to their unique structure to promote many cellular processes. In response to the growing popularity of TPMS scaffolds amongst researchers, this review discusses the effect of different parameters (including pore size, porosity, and pore shape, as well as their influences on mechanical property, permeability, and curvature), along with the controlling and designing of such parameters, on bone regeneration to serve as a guide for future researchers in designing and utilizing TPMS scaffolds for bone regeneration purposes.
Application of TPMS structure in bone regeneration
Zhifei Dong, Xin Zhao
Engineered Regeneration 2021
Bone defect repair, due to its complex process in nature, has become a costly issue in modern day medicine. This causes a growing demand for a bone substitute that is effective and easy to construct. Recently, triply periodic minimal surface (TPMS) scaffolds, which embody trabecular bone-mimicking hyperboloidal topography, have become a promising candidate for this exact role due to their unique structure to promote many cellular processes. In response to the growing popularity of TPMS scaffolds amongst researchers, this review discusses the effect of different parameters (including pore size, porosity, and pore shape, as well as their influences on mechanical property, permeability, and curvature), along with the controlling and designing of such parameters, on bone regeneration to serve as a guide for future researchers in designing and utilizing TPMS scaffolds for bone regeneration purposes.